Abstract
Allogeneic bone marrow transplantation (BMT) is the only curative treatment available for sickle cell disease (SCD). The effectiveness of the treatment is primarily assessed by the chimerism rate, which is the fraction of cells derived from the donor's hematopoietic stem cells (HSCs) relative to the recipient's cells. New less toxic conditioning agents, described as reduced, nonmyeloablative (as opposed to myeloablative conditioning agents) are now used for BMT with sibling donors. Moreover, most sibling donors are sickle cell trait carriers (heterozygous A/S genotype). In the absence of a test performed on erythroid cells, the rate of chimerism is currently assessed either on different subpopulations of myeloid cells by molecular biology techniques or on hemoglobin measurement by chromatography (HPLC). However, the percentage of myeloid chimerism is rarely representative of the percentage of erythroid chimerism in these patients and HPLC cannot allow analysis at the single cell level.
An easy-to-implement method for accurate measurement of fetal hemoglobin (HbF) content in individualized red blood cells (RBCs) has recently been developed (Hebert, et. al, AJH 2020). This method relies on flow cytometry for cell individualization and intracellular labeling of HbF with a fluorescent monoclonal antibody for quantification. Based on the same approach, we propose the use of monoclonal antibodies directed against adult hemoglobins (HbA0 and HbA2) or hemoglobin S (HbS). This strategy allows to discriminate, from a blood sample, the different RBC subpopulations coming from either the recipient or the donor, or from the transfusion associated with this procedure, thus allowing us to precisely determine the rates of erythroid chimerism in patients.
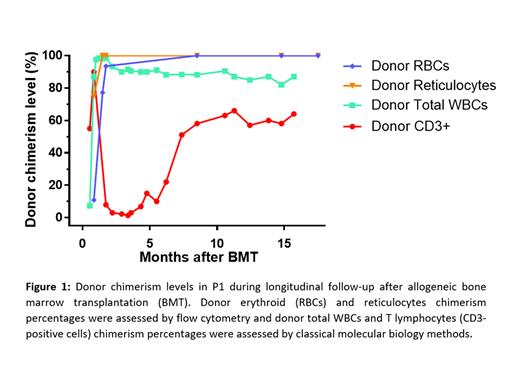
As a proof of concept, we show here the evaluation of the percentage of erythroid chimerism during a longitudinal follow-up performed on 3 patients with SCD (P1 - 18 months, P2 - 4 months, and P3 - 4 months). The 3 patients received allogeneic BMT from a sibling A/S donor following nonmyeloablative conditioning consisting of the administration of an anti-CD52 antibody associated with radiotherapy (300 cGy TBI - total-body irradiation). We observed for P1 and P2 donor erythroid chimerism >90% and >95%, 1 and 2 months after BMT, respectively (measured after exclusion of transfused RBCs). These results are in accordance with the presence of most RBCs HbA- and HbS-positive, as determined by flow cytometry. In addition, the analysis of the reticulocyte (retic) subpopulations (CD71-positive cells) of these patients provides precise information on the RBC production of the bone marrow following transplantation. Retic donor chimerism levels were >99% for both P1 and P2, 1 month after BMT procedure. P1 and P2 showed stable donor reticulocyte and donor erythroid chimerism levels during the follow-up. On the contrary, P3 presented a different response with 43%, 41% and 5% donor erythroid chimerism 1, 2 and 3 months after BMT, respectively. Retrospectively, this bad response in P3 would have been predicted by low donor reticulocyte chimerism levels (5%, 6% and 0.5% at 1, 2 and 3 months after BMT, respectively). Our erythroid specific chimerism results were also compared to the assessment of chimerism performed on either total white blood cells (WBCs) or lineage specific subpopulations after sorting. Over 18 months after BMT in P1, the correlation between retic chimerism and total WBCs or T lymphocytes (CD3-positive cells) chimerism were not statistically significant (P = 1 and P = 0.833, respectively - Spearman test) (Figure 1).
This new tool to monitor BMT outcome to treat SCD shows great precision and potentially a stronger predictive capability than total WBCs or lineage specific chimerism assessed by molecular biology. Our results highlight the importance of analyzing reticulocyte population to better assess BMT efficacy and predict the outcome a few days after the transplant. Its use could also be considered in the context of the various gene therapy strategies currently under development using modified β-globin gene addition or direct mutation correction through CRISPR/Cas9 and homologous recombination system.
Bencheikh: Innovhem: Current Employment. Bartolucci: Innovhem: Current holder of individual stocks in a privately-held company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal